Many of these are reviewed below in their order in the Periodic Table: The incorporation of other transition metals into steel has strengthening and resistance effects that further improve the suitability of different steels for structural and non-structural applications. 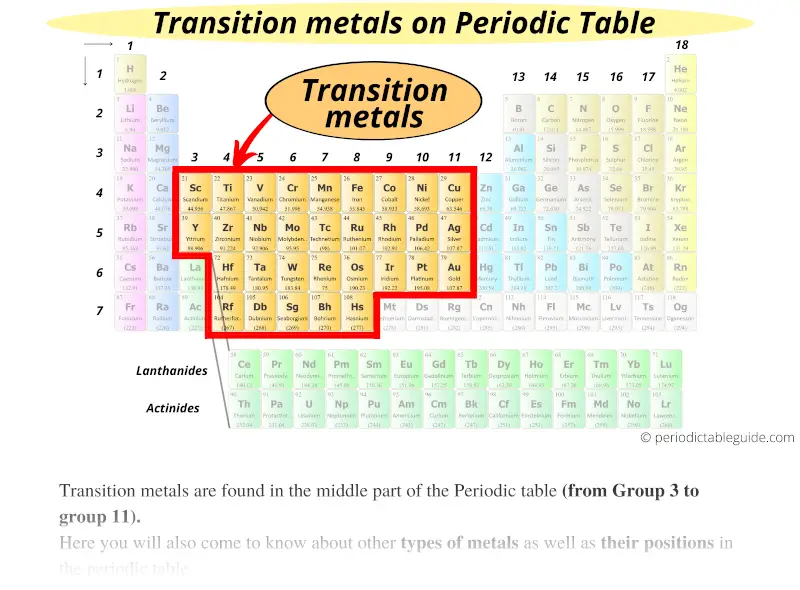
For iron this is the conversion from Fe(0) to the iron (III) oxidation state, which is explained more here (link to corrosion page) The natural tendency for pure metals to revert to their native (ore) oxidation state represents the continuous battle for industry against the effects of corrosion. 
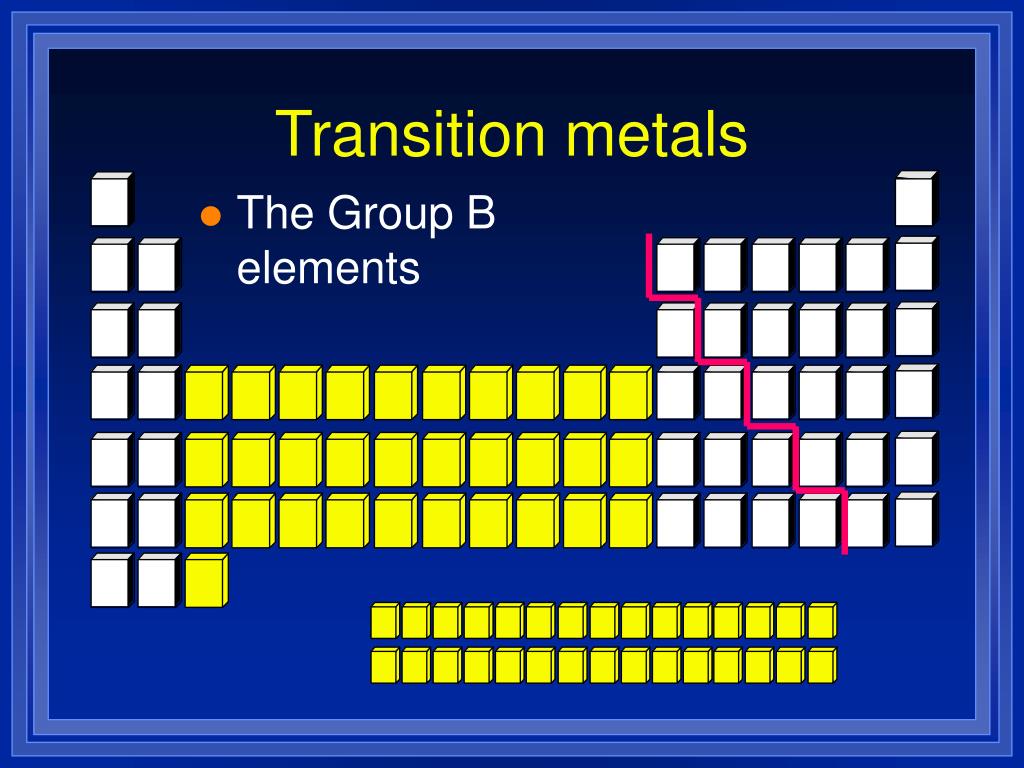
Iron is arguably the most well-known transition metal for its biological necessity and as the principal element in steel, which is an alloy of iron and carbon. Transition metals and corrosion mitigation Transition metals also have high melting points and boiling points when compared to the softer alkali metals of the Periodic Table. The whole set of transition metals can be seen in the Periodic Table below:Ĭorrosionpedia Explains Transition MetalsĪs a result of their d-shell electronic structures, the transition metals exhibit many different chemical properties including the formation of co-ordination compounds (complexes) as well as functioning as catalysts through their oxidation-reduction behavior. In order of increasing atomic mass and appearance in the table, these are: scandium, titanium, vanadium, chromium, manganese, iron, cobalt, nickel, copper and finally zinc. The top row ('the 3d elements') contains metallic elements that are most familiar. There are four rows of transition metals in the Periodic Table. Transition metals also form alloys, which is significant in corrosion mitigation, and are ductile and malleable too. They don't react readily with water and oxygen (unlike the alkali metals of the Periodic Table) but will react with oxygen at elevated temperatures. Transition metals in their pure form are generally corrosion resistant, owing to the fact that they can form stable oxide layers on the surface. They are defined by their electronic structures incorporating d-shell electrons and they are known for their ability to exhibit and move between different oxidation states as a result. Those two orbitals can use the electrons to bond with other atoms.A transition metal is one of a group of 40 metallic chemical elements in the Periodic Table. Example: Molybdenum (Mo), with 42 electrons. Whenever you have a shell that is not happy, the electrons want to bond with other elements. Why can they do that? As you learn more, you will discover that most transition elements actually have two shells that are not happy. It's a chemical trait that allows them to bond with many elements in a variety of shapes. Transition metals can use the two outermost shells/orbitals to bond with other elements. Most elements can only use electrons from their outer orbital to bond with other elements. You will find it's usually 2, 8, 18 or 32 for the maximum number of electrons in an orbital. No shell can have more than 32 electrons. Something like gold (Au), with an atomic number of 79, has an organization of 2-8-18-32-18-1. The transition metals are able to put up to 32 electrons in their second-to-last shell. You need to remember that those electrons are added to the second-to-last shells. This is the point in the periodic table where you can place more than 8 electrons in a shell. Scandium (Sc) is only 3 spots away with 21 electrons, but it has a configuration of 2-8-9-2. It has 18 electrons set up in a 2-8-8 order. Transition metals are able to put more than eight electrons in the shell that is one in from the outermost shell. Not all of them, but we are sure you've seen pictures of silver (Ag), gold (Au), and platinum (Pt). You will usually find that transition metals are shiny, too. They have a lot of electrons and distribute them in different ways. Transition metals are good examples of advanced shell and orbital ideas. We like introducing students to the first eighteen elements, because they are easier to explain. It all has to do with their shells/orbitals. Let's start off by telling you that there are a lot of elements that are considered transition metals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
